Learning check
Once you have watched the video, check your learning with this quiz.
When was chemistry born?
Was chemistry born when metallurgy started to develop?
The bronze age (3200 – 500 BC):
Was chemistry born when the first philosophers pondered upon what materia was?
Thales of Miletus (625 – 545 BC)
- “Father of philosophy”
- “Water is the permanent entity.”
- It can change form …
- No experiments!
Empedocles (490 – 434 BC)
- Showed that air is not empty space!
- Put your finger on top of a straw.
- Place the straw in a glass of soda.
- The soda doesn't enter the straw until you remove your finger ⇒ air is some kind of matter!
- “All matter is made up of water, air, earth, and fire.”
Democritus (460 – 370 BC)
- Proposes the first “atomic theory”.
- “Atomos” in Greek means “indivisible”.
Plato (428 – 348 AD) and his student Aristotle (384 – 322 AD)
- Favor the idea of four (or five) elements.
- The idea of the four elements persists for 2000 years …
The age of alchemy
Was chemistry born when alchemists first tried to transmute metals?
Middle ages
- Europe’s monks give science “artificial ventilation”.
- Science instead flourishes in the Arabic world.
- Abu Mūsā Jābir ibn Hayyān (“Geber”).
Hennig Brand: The last alchemist
- In search of “the philosopher’s stone”.
- Discovers phosphorus 1669.
The fathers of modern chemistry
Robert Boyle: The first chemist
- “The Sceptical Chymist” (1661).
- Gets rid of “al-” in “alchemy”, and also the magical/mystical part.
- Chemistry must be based on experiments!
Boyle's experiments
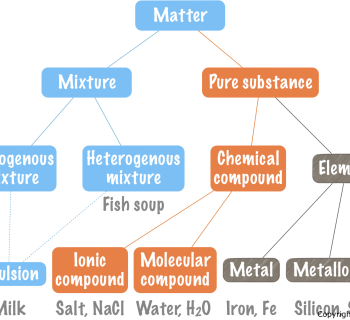
- Distinguishes mixtures from compounds.
- Experiments with gases: Sound requires a medium.
- Substances consist of particles with different sizes.
The discovery of oxygen
- Joseph Priestley publishes his findings on "flammable air" 1175.
- Antoine Lavoisier realizes the role of oxygen in combustions, gives oxygen its name.
- (Lavoisier also discovers hydrogen, the conservation of mass, constructs the metric system, writes list of elements …)
Jöns Jacob Berzelius develops the chemical language.
- Gives the chemical elements their symbols: H, C, N, O, Fe etc. (1813).
- Introduces chemical formulas.
- Coins the terms catalysis, polymer, protein …
- Compiles a list of atomic weights of the known elements.