Learning check
Once you have watched the video, check your learning with this quiz.
A sodium atom is oxidized
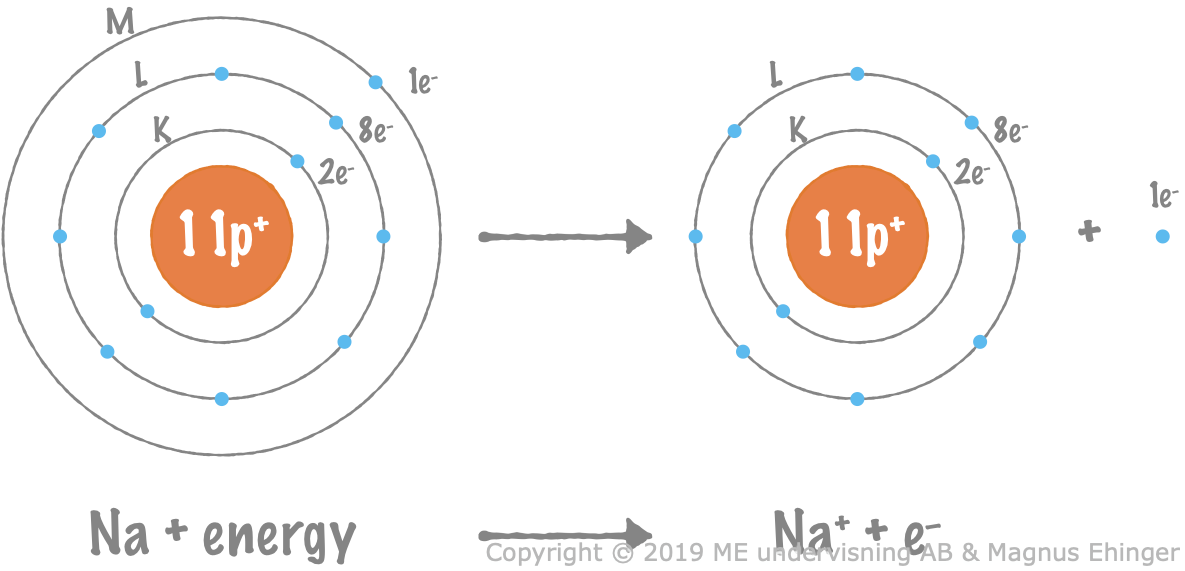 When a sodium atom is oxidized, it gives off an electron and turns into a sodium ion, Na+.
When a sodium atom is oxidized, it gives off an electron and turns into a sodium ion, Na+.
Energy is required to remove the valence electron from the sodium atom.
- A sodium ion, Na+, is formed, as an electron is released.
- The sodium atom now has eight valence electrons (noble gas configuration), which is particularly stable.
A chlorine atom is reduced
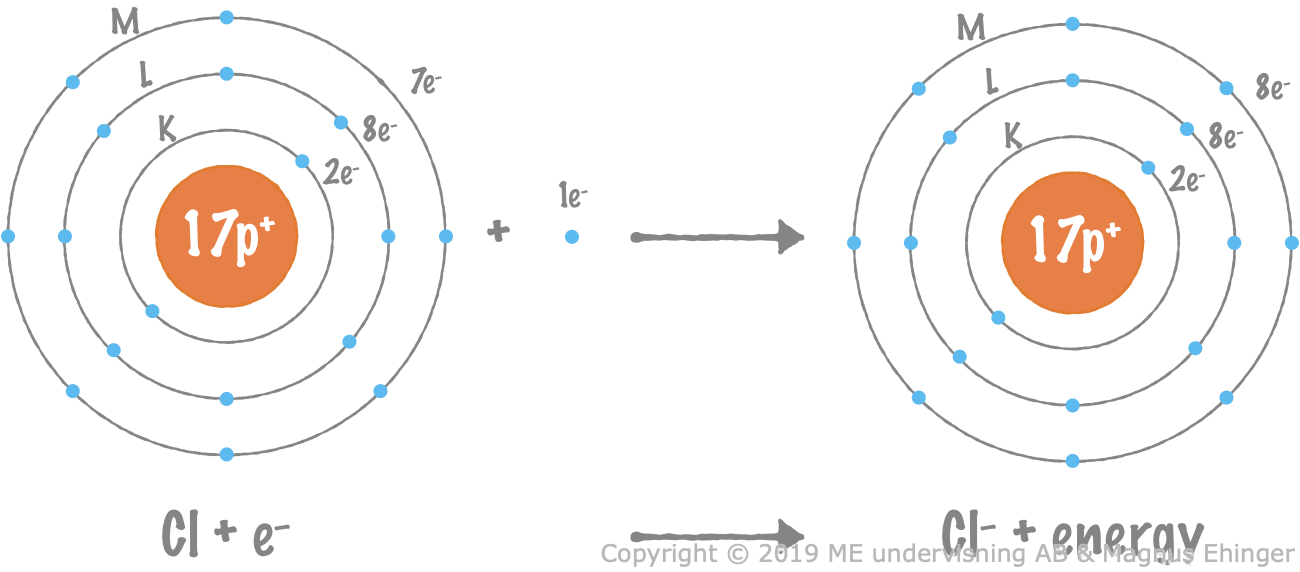 When a chlorine atom is reduced, it takes up an electron and turns into a chloride ion, Cl–.
When a chlorine atom is reduced, it takes up an electron and turns into a chloride ion, Cl–.
Energy is released when a chlorine takes up an extra electron.
- A chloride ion, Cl–, is formed.
- The chloride ion now has eight valence electrons (noble gas configuration), which is particularly stable.
A redox reaction
With words:
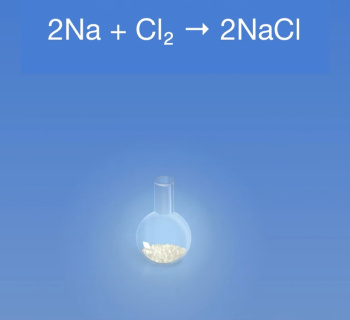
- Sodium + chlorine gas → sodium chloride
With Lewis structures:
-
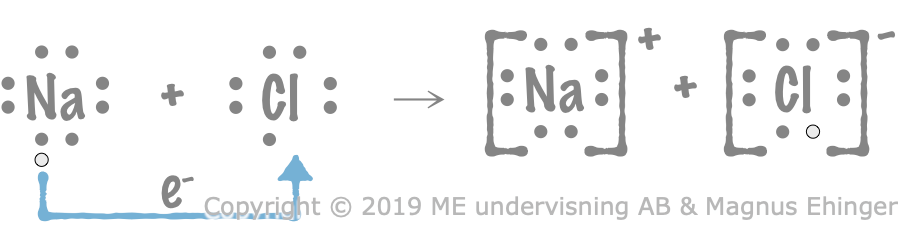 When sodium is oxidized by chlorine, both ions that form get eight valence electrons (noble gas configurations).
When sodium is oxidized by chlorine, both ions that form get eight valence electrons (noble gas configurations).
With electron configurations:
-
K L M K L M K L M K L M 11p+ 2e– 8e– 1e– + 17p+ 2e– 8e– 7e– → 11p+ 2e– 8e– 0 + 17p+ 2e– 8e– 8e– sodium atom, Na chlorine atom, Cl sodium ion, Na+ chlorine ion, Cl–
Oxidation + reduction = redox reaction
We can combine the oxidation reaction and the reduction reaction to a redox reaction by adding the two together.
| Oxidation: | Na → Na+ + e– | ×2 | |
| + | Reduction: | Cl2 + 2e– → 2Cl– | |
| Redox: | 2Na + Cl2 → 2NaCl | ||
Chlorine gas consists of chlorine molecules, Cl2. Because there are two chlorine atoms in each chlorine molecule, two electrons are taken up by each chlorine molecule. Because of this, the oxidation reaction must be taken ×2 before we can add the reactions together.
- The two e– that are given off in the oxidation reaction are taken up in the reduction. Because of this, we don't need to write them in the redox reaction formula.