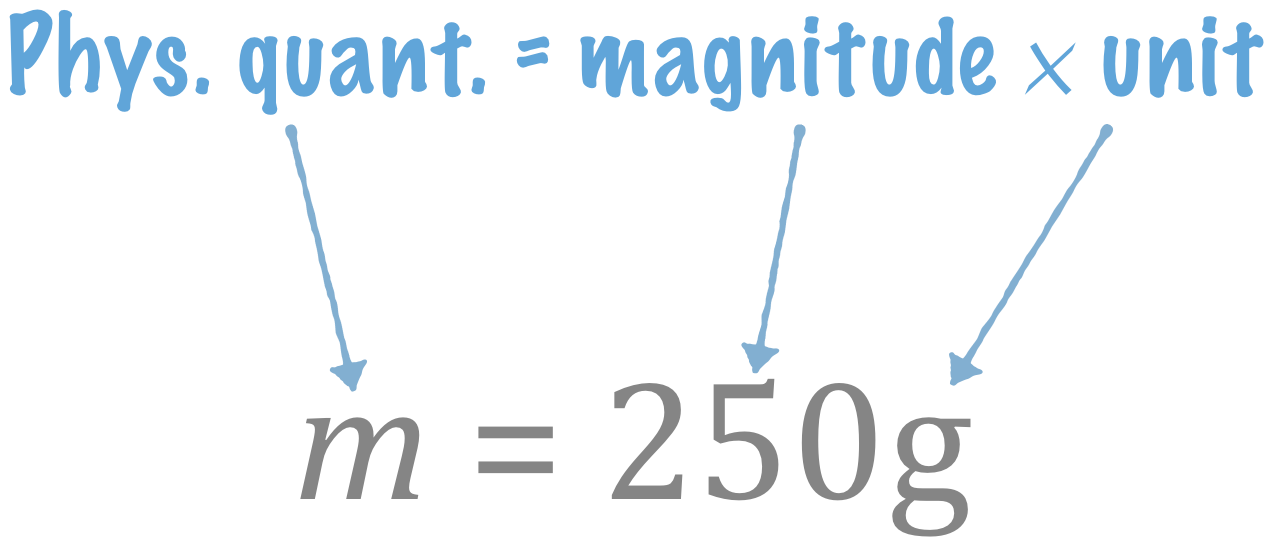[Sorry, the video "Physical quantity, magnitude, and units" has not yet been published.]
Learning check
[Sorry, the learning check for this video has not yet been published.]
Physical quantity
A physical quantity is something you can measure.
Examples:
- Length, \(l\)
- Weight (mass), \(m\)
- Speed, \(v\)
- Voltage, \(U\)
- Cost
Unit
A unit is a definite magnitude of a certain physical quantity.
Examples:
- Length, \(l\) – meter/metre, \(\text{m}\)
- Weight (mass), \(m\) – gram, \(\text{g}\)
- Speed, \(v\) – meters per second, \(\text{m/s}\)
- Voltage, \(U\) – volt, \(\text{V}\)
- Cost – € (Euro) etc.
SI units
Get used to them!
- Mass: 1 kg = 1000 g
- Volume: 1 L = 1 dm3 = 1000 mL
Also: Learn the prefixes:
| Prefix | Base 10 | |
| Name | Symbol | |
| mega | M | 106 |
| kilo | k | 103 |
| deci | d | 10–1 |
| milli | m | 10–3 |
| micro | μ | 10–6 |
Numerical magnitude
The amount (magnitude) of the unit you’re measuring.
Examples:
| Phys. quant. | Example | Numerical magnitude |
| Length | \(l = 1.93\text{m}\) | 1.93 |
| Mass | \(m = 250\text{g}\) | 250 |
| Speed | \(v = 25\text{m/s}\) | 25 |
| Voltage | \(U = 12\text{V}\) | 12 |
The relation between physical quantity, magnitude, and unit
Note: There is a multiplication sign between "\(250\)" and "\(\text{g}\)": \(m = 250 \times \text{g}\)
- This is similar to algebraic notation, e.g. \(y = 250x\).
How to use physical quantity, magnitude, and unit
Example 1
If I dissolve 25g salt in 0.5dm3 water, which is the salt concentration? Give your answer in the unit g/dm3.
Solution
Since the answer is to be given in the unit \(\frac {\text{g}}{\text{dm}^3}\), I have to divide the mass \(m\) by the volume \(V\). We write the concentration \(c\):
\[c = \frac {m}{V} = \frac {250\text{g}}{0.5\text{dm}^3} = 50 \frac {\text{g}}{\text{dm}^3}\]
Answer: \(c = 50\text{g/dm}^3\)
↑ Note: Both physical quantity, magnitude, and unit in the answer!
Example 2
A salt solution has a concentration of 50g/dm3. From this solution I pour 0.100dm3 in a glass. What is the mass of the salt in the glass?
Solution
I want to know the mass \(m\), which is measured in \(\text{g}\).
I know the concentration \(c\), which is measured in \(\frac {\text{g}}{\text{dm}^3}\).
How do we go from the unit \(\frac {\text{g}}{\text{dm}^3}\) to \(\text{g}\)? We must multiply \(\frac {\text{g}}{\text{dm}^3}\) with \(\text{dm}^3\):
\[\frac {\text{g}}{\text{dm}^3} \times \text{dm}^3 = \text{g}\]
Thus, we can write:
\[m = cV = 500\frac {\text{g}}{\text{dm}^3} \times 0.100\text{dm}^3 = 50.0\text{g}\]
Answer: \(m = 50.0\text{g}\)