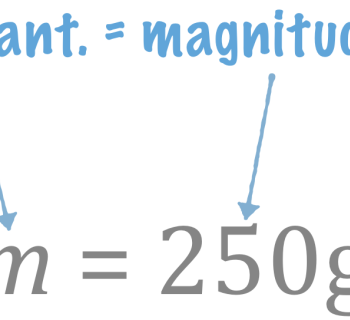Learning check
Once you have watched the video, check your learning with this quiz.
The mole
What's the mass of one molecule of H2?
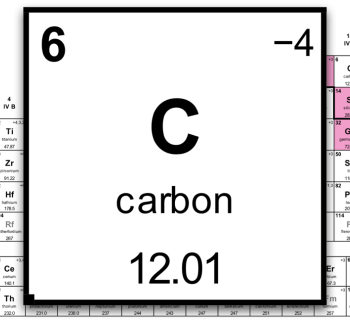
- Let’s have a look at the periodic table!
- One hydrogen atom weighs 1.008 u.
- One hydrogen molecule, H2, weighs 2 × 1.008u = 2.016u.
 Hydrogen is up to the left of the periodic table, and oxygen is up to the right.
Hydrogen is up to the left of the periodic table, and oxygen is up to the right.
What’s the mass of 6.022 × 1023 H2-molecules?
- 1 u is the equivalent of 1.6605 × 10–24 g
- A single H2-molecule weighs 2.016 u =
= 2.016 u × 1.6605 × 10–24 g/u = 3.347568 × 10–24 g - 6.022 × 1023 H2-molecules weigh 6.022 × 1023 × 3.347568 × 10–24 g =
= 2.01590545 g ≈ 2.016 g
What’s the mass of one molecule of O2?
- Let’s have a look at the periodic table again!
- One oxygen atom weighs 16.00 u.
- One oxygen molecule, O2, weighs 2 × 16.00u = 32.00u.
What’s the mass of 6.022 × 1023 O2-molecules?
- 1 u is the equivalent of 1.6605 × 10–24 g
- A single O2-molecule weighs 32.00 u =
= 32.00 u × 1.6605 × 10–24 g/u = 5.3136 × 10–23 g - 6.022 × 1023 O2-molecules weigh 6.022 × 1023 × 5.3136 × 10–23 g =
= 31.9984992 g ≈ 32.00 g
| 1 molecule | weighs | 6.022 × 1023 molecules weigh |
| H2 | 2.016 u | 2.016 g |
| O2 | 32.00 u | 32.00 g |
6.022 × 1023 = 1 mol
- ”Mole” (symbol: mol) is a ”word of quantity”
- More ”words of quantities”:
- 1 pair (couple) = 2 pcs
- 1 dozen = 12 pcs
- score = 20 pcs
- gross = 144 pcs
- grand = 1,000 pcs
- myriad = 10,000 pcs
- 1 mol = 6.022 × 1023 pcs
How do you use the number 6.022 × 1023?
- The number 6.022 × 1023 is a conversion factor from u → g
- The number 6.022 × 1023 is called Avogadro’s constant. It’s written \(N_\text{A}\) and has the unit \(1/\text{mol}\).
- We write: \(N_{\text{A}} = 6.022 \times 10^{23}/\text{mol}\)
Amount of substance
The amount of substance answers the question, ”How many moles of the substance is there?”
- Written \(n\), has the unit mol.
- Example: \(n = 25 \text{mol}\)
Example 1I have a piece of iron that I know contains 2.5 moles of iron atoms. How many iron atoms is that? Solution\(N_{\text{Fe}} = n_{\text{Fe}} \times N_{\text{A}} =\) \(2.5\text{mol} \times 6.022 \times 10^{23}/\text{mol} = 1.5055 \times 10^{24} \approx 1.5 \times 10^{24}\) |
Example 2A piece of gold that I have in my lab consists of 6.1 × 1021 gold atoms. What is the amount of substance of gold, i.e. how many moles of gold are there? Solution\(n_{\text{Au}} = \frac {N_{\text{Au}}}{N_{\text{A}}} = \frac {6.2 \times 10^{21}}{6.022 \times 10^{23}/\text{mol}} = 0.0101295\text{mol} \approx 10 \times 10^{-3}\text{mol}\) |
Molar mass
1 mole of something is 6.022 × 1023 pcs.
- The molar mass of something tells us how much one mole of that substance weighs.
- The molar mass is written \(M\) and has the unit \(\text{g/mol}\).
What is the molar mass of hydrogen gas, H2?
\(M_{\text{H}_2} = m_{\text{1 H}_2\text{ molecule}} \times N_{\text{A}}\)
\(m_{\text{1 H}_2\text{ molecule}} = 2.016\text{u} \times 1.6605 \times 10^{-24}\text{g/u} = 3.347568 \times 10^{-24}\text{g}\)
\(M_{\text{H}_2} = 3.347568 \times 10^{-24}\text{g} \times 6.022 \times 10^{23}/\text{mol} ≈ 2.016\text{g/mol}\)
A mathematical relation
The molar mass indicates how much 1 mol of something weighs.
- Thus, we can write:
\[\text{molar mass} = \frac{\text{mass}}{\text{amount of substance}}\]
- Or, preferably:
\[M = \frac{m}{n}\]
Where
\(M\) is the molar mass in \(\text{g/mol}\).
\(m\) is the mass in \(\text{g}\).
\(n\) is the amount of substance in \(\text{mol}\).
Example 3What is the amount of substance in 36.0 g water? SolutionLet’s use our mathematical relation: \[M = \frac {m}{n} \Leftrightarrow n = \frac {m}{M}\] \(n_{\text{H}_2\text{O}} = \frac {m_{\text{H}_2\text{O}}}{M_{\text{H}_2\text{O}}}\) \(m_{\text{H}_2\text{O}}\) \(M_{\text{H}_2\text{O}} = 1.008\text{g/mol} \times 2 + 16.00\text{g/mol} = 18.016\text{g/mol}\) \(n_{\text{H}_2\text{O}} = \frac {36.0\text{g}}{18.016\text{g/mol}} = 1.9982238\text{mol} \approx 2.00\text{mol}\) |