Learning check
Once you have watched the video, check your learning with this quiz.
Reactions may go both ways
Most of the reactions we have looked at so far go completely to the right: All the reactants are converted into products. This is the case in many redox reactions.
- Combustion of sugar:
C12H22O11 + 12O2 → 12CO2 + 11H2O - Dissolving magnesium in hydrocloric acid:
Mg(s) + 2HCl(aq) → H2(g) + MgCl2(aq)
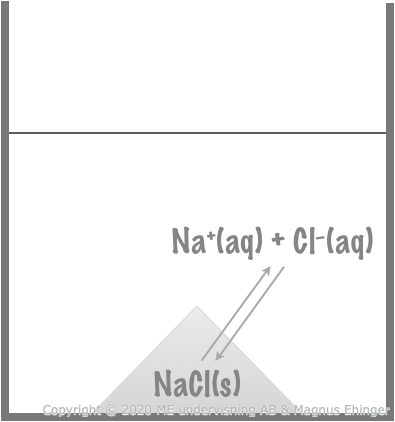
In a saturated table salt solution, there are two reactions balancing each other.
- Saturated = no more salt can be dissolved, and a solid precipitate of NaCl(s) is formed.
One reaction:
- NaCl(s) → Na+(aq) + Cl–(aq)
At exactly the same rate, the opposite reaction takes place:
- Na+(aq) + Cl–(aq) → NaCl(s)
The system is at equilibrium, because:
- Na+(aq) + Cl–(aq) is formed at exactly the same rate as NaCl(s).
- The reaction to the right is exactly as fast as the reaction to the left: \(\overrightarrow{v} = \overleftarrow{v}\)
What happens when a reaction reaches equilibrium?
An imaginary reaction: A + B ⇌ C + D
We start with some concentrations A and B, and no C and D at all.
- Because [A] and [B] are quite large, the reaction rate to the right, \(\overrightarrow{v}\) is quite high.
- Because [C] and [D] equal zero, \(\overleftarrow{v} = 0\).
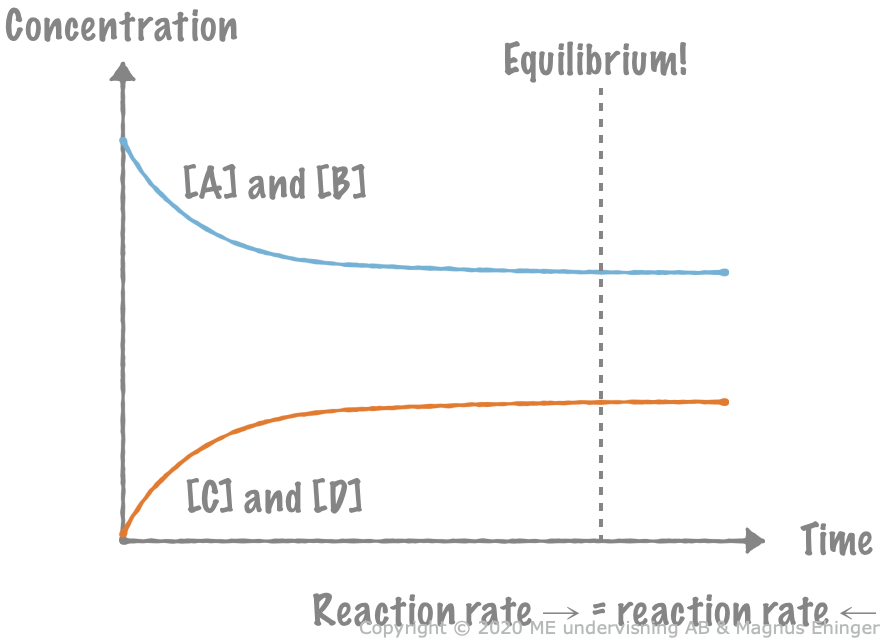 The reaction A + B ⇌ C + D reaches equilibrium.
The reaction A + B ⇌ C + D reaches equilibrium.
As some C + D is formed, \(\overleftarrow{v}\) increases. At the same time, [A] and [B] decrease, which causes \(\overrightarrow{v}\) to decrease, too.
At some point, \(\overleftarrow{v}\) has increased so much, and \(\overrightarrow{v}\) has decreased so much, that \(\overrightarrow{v} = \overleftarrow{v}\).
- The two reactions, to the left and to the right, balance each other perfectly.
- From this point forward, the system is at equilibrium.
At equilibrium, the concentrations remain constant
[A], [B], [C], and [D] don’t change any more.
- Expressed mathematically:
\[K= \frac{[\text{C}][\text{D}]}{[\text{A}][\text{B}]}\]
- \(K\) is called the equilibrium constant.
- The expression above is called the law of mass action or the Guldberg-Waage law.
What affects the value of the equilibrium constant \(K\)?
For a given reaction:
- Only the temperature
- NOT the initial concentrations or concentration change
- NOT any catalysts
- Only the temperature