Learning check
Once you have watched the video, check your learning with this quiz.
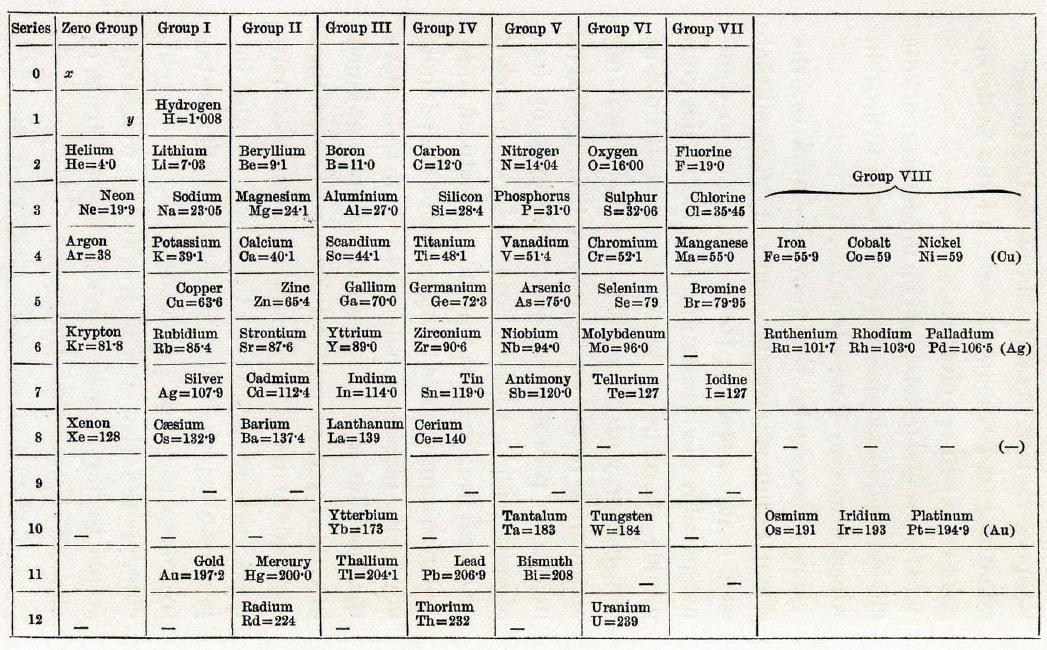
The periodic table
Middle of the 19th century: Some 60 elements discovered.
- Mendeleev organizes them by atomic mass.
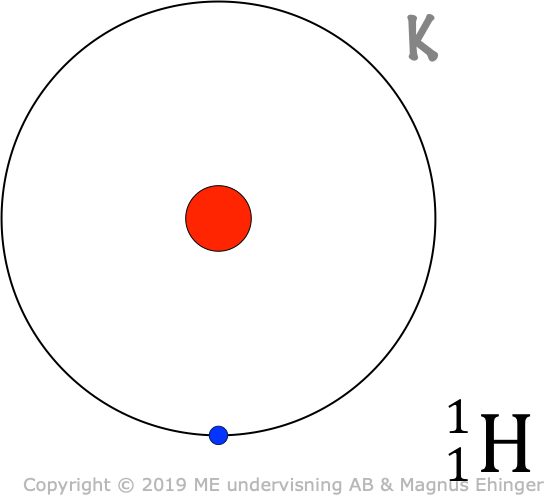
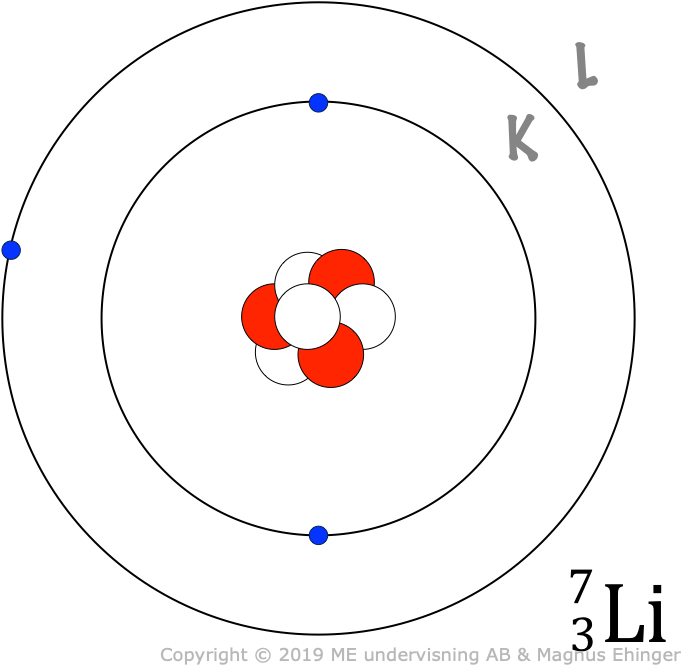
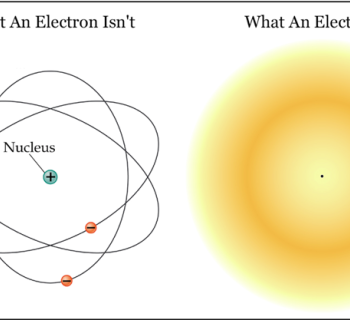
Models of some atoms
How many e– are there room for in the different shells?
| Shell | 1 (K) | 2 (L) | 3 (M) | 4 (N) | \(n\) |
| Max. number of e– | 2 | 8 | 18 | 32 | \(2n^2\) |
Electron configurations for some atoms
All the electrons marked in red are valence electrons.
| Element | Atomic # | K | L | M | N |
| Hydrogen, H | 1 | 1 |
|
|
|
| Helium, He | 2 | 2 |
|
|
|
| Litium, Li | 3 | 2 | 1 |
|
|
| Beryllium, Be | 4 | 2 | 2 |
|
|
| Fluorine, F | 9 | 2 | 7 |
|
|
| Neon, Ne | 10 | 2 | 8 |
|
|
| Sodium, Na | 11 | 2 | 8 | 1 |
|
| Sulfur, S | 16 | 2 | 8 | 6 |
|
| Argon, Ar | 18 | 2 | 8 | 8 |
|
| Potassium, K | 19 | 2 | 8 | 8 | 1 |
| Zinc, Zn | 30 | 2 | 8 | 18 | 2 |
| Krypton, Kr | 36 | 2 | 8 | 18 | 8 |
Example
Give the electron configuration for oxygen, 8O.
Solution
Oxygen has 8p+ and 8e–
| K | L | M | |
| 8p+ | 2e– | 6e– | 0 |
Valence electrons
Valence electrons decide much of an element's properties.
- These properties were what Mendeleev noticed!
- The farther away from the nucleus, the easier they may be released.
- The fewer they are, the easier they may be released.
- Eight valence electrons are particularly stable!
Rule of octets – noble gas configuration
- All the noble gases except helium have eight valence electrons.
- Eight valence electrons (or two, for helium) is called noble gas configuration.
- In chemical reactions: The atoms strive to attain noble gas configuration.